Scientists say an old Epilepsy drug could transform sleep apnea treatment with a simple nightly pill
Epilepsy drug, Sultiame trial shows promise for sleep apnea patients
A decades-old epilepsy drug may be on the verge of reshaping how doctors treat obstructive sleep apnea (OSA), one of the world’s most common and under-treated sleep disorders. New clinical trial findings suggest that sultiame (also known as sulthiame), a medication historically used for certain seizure disorders in Europe, significantly reduced breathing interruptions during sleep in patients with moderate to severe OSA.
The findings, published in The Lancet, are drawing attention because they hint at something patients and clinicians have wanted for years: a pill-based alternative to CPAP machines. For millions who struggle to tolerate masks, tubing, and pressurized airflow, this could mark a major step toward a simpler treatment option.
A Potential Sleep Apnea Breakthrough: Epilepsy Drug Sultiame Shows Strong Trial Results
Researchers in a multicenter Phase II clinical trial tested sultiame in 298 adults with untreated moderate to severe obstructive sleep apnea. According to the study summaries provided by Gizmodo and ScienceDaily, participants were randomized to receive either a placebo or different doses of the drug over a 15-week period.
The most striking result: patients on higher doses of sultiame experienced up to a 47% reduction in breathing interruptions, also known as apnea events. That level of improvement was paired with better oxygen levels during sleep, improved sleep quality, and reduced daytime sleepiness.
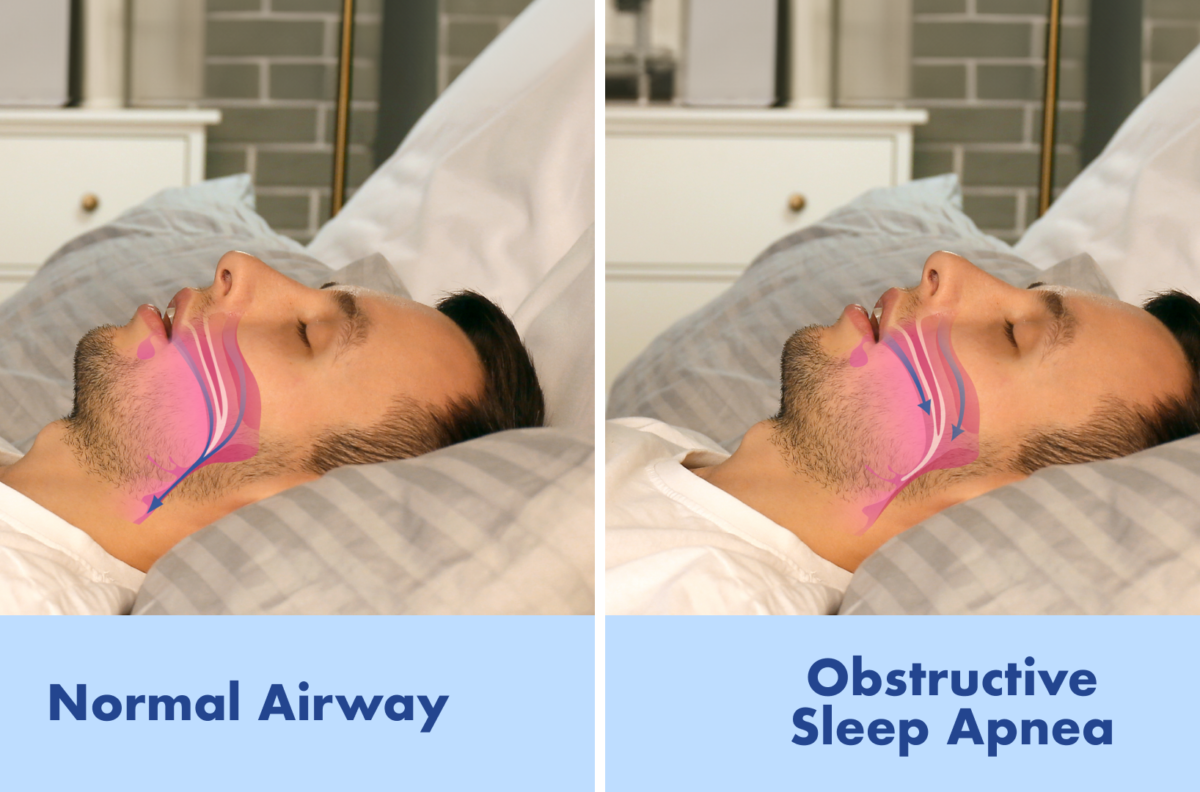
This is significant because OSA is not just about snoring or restless sleep. Repeated airway collapse during the night can lower oxygen levels and place long-term stress on the body, increasing the risk of high blood pressure, cardiovascular disease, stroke, type 2 diabetes, and possibly cognitive decline.
What Is Sultiame and Why Is an Epilepsy Drug Being Studied for Sleep Apnea?
Sultiame is not a new drug. It was first introduced in the 1960s as an anti-seizure medication and is still used in parts of Europe for epilepsy treatment, especially certain pediatric seizure disorders. However, scientists now believe its mechanism may make it useful far beyond neurology.
The drug works by inhibiting carbonic anhydrase, an enzyme involved in the body’s regulation of breathing. In practical terms, researchers believe sultiame helps stabilize respiratory control and increase respiratory drive, reducing the chance that the upper airway collapses during sleep — the core problem in obstructive sleep apnea.
That is why this study matters so much. Rather than merely addressing symptoms, sultiame appears to target a key biological driver of sleep apnea episodes.
Why a Pill for Sleep Apnea Could Be a Game-Changer Beyond CPAP
For years, CPAP (continuous positive airway pressure) has been considered the gold-standard treatment for obstructive sleep apnea. It is highly effective when used consistently, because it physically keeps the airway open with pressurized air delivered through a mask.
But there is a catch: many patients simply cannot tolerate CPAP long-term. Masks can feel uncomfortable, noisy, restrictive, or disruptive to sleep. According to the provided source material, up to half of patients stop using CPAP within a year.
That reality has created enormous demand for alternatives. A daily pill could dramatically improve adherence for patients who abandon CPAP or never start it in the first place. If future trials confirm these results, sultiame may become one of the most closely watched candidates in the growing race to develop non-device sleep apnea treatments.
What the Researchers Said About the Sleep Apnea Pill Trial
The study’s investigators described the findings as highly encouraging. In comments highlighted by the University of Gothenburg and echoed in the media coverage, Jan Hedner, senior professor of pulmonary medicine, said the results suggest that sleep apnea “can indeed be influenced pharmacologically.”
He also emphasised caution, noting that larger and longer studies are still needed to determine whether the benefits hold up over time and whether the treatment remains safe across broader patient populations.
That balance is important. While the current results are promising, this is still a Phase II trial, which means the research is not yet at the point where doctors can broadly prescribe sultiame specifically for OSA in routine clinical practise.
READ ALSO
Is Sultiame Approved for Sleep Apnea Yet? Here’s What Patients Should Know
At this stage, the answer is no.
Sultiame is not yet approved as a sleep apnea treatment, and the article material notes that the drug was never approved in the United States for its original epilepsy use. That means U.S. patients should not expect immediate access to it as an OSA prescription.
However, the trial’s success could accelerate the next steps: larger Phase III studies, longer safety follow-up, and eventual regulatory submissions if the data continues to hold up.
For now, sultiame remains a promising experimental option, not a replacement for currently approved therapies.
The Bigger Picture: New Sleep Apnea Drugs Are Already Changing the Market
Sultiame is not the only medication drawing attention in the sleep apnea space. The source material also notes that in late 2024, Eli Lilly’s tirzepatide gained expanded approval to treat sleep apnea in certain contexts, particularly where obesity is a major driver of the condition.
That matters because it signals a broader shift: sleep apnea treatment is moving from a device-only era toward a future where multiple drug-based options may exist. Some medications may help by reducing body weight and improving airway mechanics indirectly, while others, like sultiame, may target breathing control and airway collapse more directly.
In other words, the future of sleep apnea care may be personalized, with different treatments matched to different patient profiles.
What This Means for People Living With Obstructive Sleep Apnea
For people diagnosed with obstructive sleep apnea, especially those who struggle with CPAP, the sultiame data offers something powerful: hope.
A pill that meaningfully cuts apnea events, improves oxygen levels, and reduces daytime fatigue could make treatment more accessible and easier to stick with. That said, experts are clear that patients should not stop CPAP or change treatment plans without medical guidance.
Right now, the best takeaway is this: the search for a true sleep apnea pill is no longer theoretical. With sultiame’s latest results, it is becoming a real possibility.
As more data emerges, this old epilepsy drug could become one of the most important stories in sleep medicine this year.
FAQ: Epilepsy Drug for Sleep Apnea (Sultiame)
1) Is there finally a pill for sleep apnea?
Not yet, but sultiame (sulthiame) is one of the strongest pill-based candidates seen so far. In a new Phase II trial, the drug reduced breathing interruptions in people with moderate to severe obstructive sleep apnea by up to 47% at higher doses. It is still experimental for sleep apnea and has not been approved for this use yet.
2) What is the name of the epilepsy drug being tested for sleep apnea?
The drug is called sultiame, also spelled sulthiame. It is an older anti-seizure medication that has been used in parts of Europe, particularly for certain forms of epilepsy.
3) How effective was sultiame in the sleep apnea study?
In the reported European clinical trial, patients on higher doses of sultiame had up to 47% fewer breathing interruptions during sleep compared with placebo. Researchers also reported better oxygen levels, improved sleep quality, and less daytime sleepiness.
4) Can sultiame replace CPAP machines?
Not yet. CPAP remains the standard treatment for obstructive sleep apnea because it is highly effective when patients can tolerate it. Sultiame is still being studied and is not approved as a replacement. However, it may eventually become an option for people who cannot tolerate CPAP or need an additional therapy.
5) How does sultiame work for obstructive sleep apnea?
Researchers believe sultiame helps by stabilizing the brain’s control of breathing and increasing respiratory drive. It also inhibits carbonic anhydrase, which may reduce the chance of the upper airway collapsing during sleep, the main cause of obstructive sleep apnea.
6) Is sultiame approved in the United States?
Based on the source material, sultiame has never been approved in the U.S. for its original epilepsy use, and it is not approved for sleep apnea at this time. More studies and regulatory review would be needed before it could reach the U.S. market.
7) What kind of sleep apnea does sultiame treat?
The study focused on obstructive sleep apnea (OSA), which is the most common form of sleep apnea. OSA happens when the upper airway repeatedly collapses during sleep, causing pauses in breathing and reduced oxygen levels.
8) Who was included in the sultiame sleep apnea trial?
The trial included 298 adults with untreated moderate to severe obstructive sleep apnea. It was a multicenter, randomized, double-blind, placebo-controlled, dose-finding Phase II trial conducted across four European countries.
9) What were the side effects of sultiame in the study?
According to the source material, most side effects were mild and temporary. However, detailed safety data will need to be confirmed in larger and longer-term studies before the drug can be considered for widespread use in sleep apnea patients.
10) Why are researchers excited about a sleep apnea pill?
Because many patients stop using CPAP due to discomfort, inconvenience, or intolerance. A simple nightly pill could improve adherence, expand access to treatment, and give doctors more options for tailoring care.
11) Are there other medications for sleep apnea besides sultiame?
Yes. The source material notes that tirzepatide has also gained attention in sleep apnea treatment, particularly when obesity contributes to the condition. Some newer drugs are being developed to target airway collapse, respiratory control, or weight-related causes of OSA.
12) When could a sleep apnea pill become available?
There is no confirmed launch date. Sultiame still needs larger Phase III trials, longer safety monitoring, and regulatory review. If future data remains positive, it could eventually move toward approval, but patients should expect the process to take time.
13) Should people with sleep apnea stop using CPAP and wait for sultiame?
No. Anyone currently using CPAP or another prescribed therapy should continue their treatment unless their doctor advises otherwise. Sultiame is still experimental for sleep apnea and should not be viewed as a currently available substitute.
14) Why is sleep apnea considered dangerous?
Untreated obstructive sleep apnea can increase the risk of high blood pressure, heart disease, stroke, type 2 diabetes, and chronic daytime fatigue. It may also affect memory, concentration, and overall quality of life.
15) What makes this sleep apnea study important?
This trial is important because it suggests that sleep apnea may be treatable with a targeted medication, not just machines or surgery. If confirmed in later trials, sultiame could help usher in a new era of personalized, pill-based sleep apnea care.

